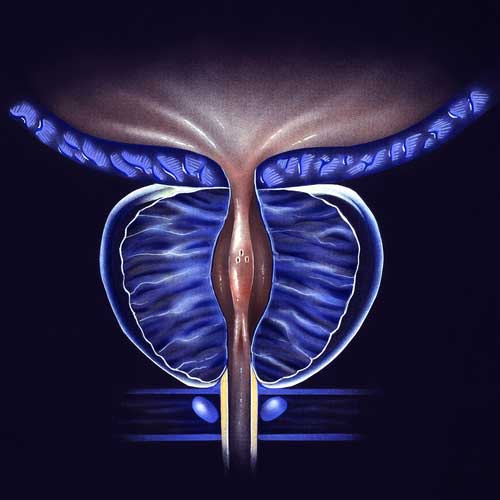
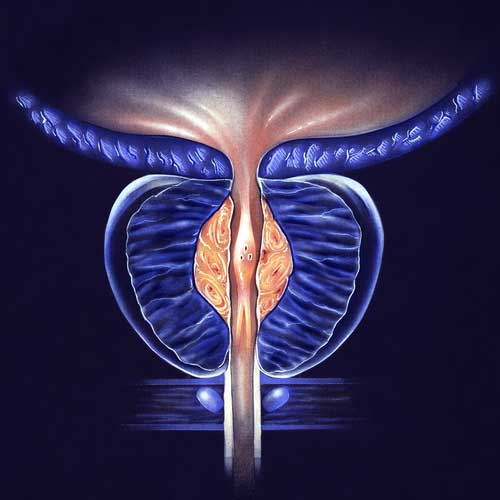
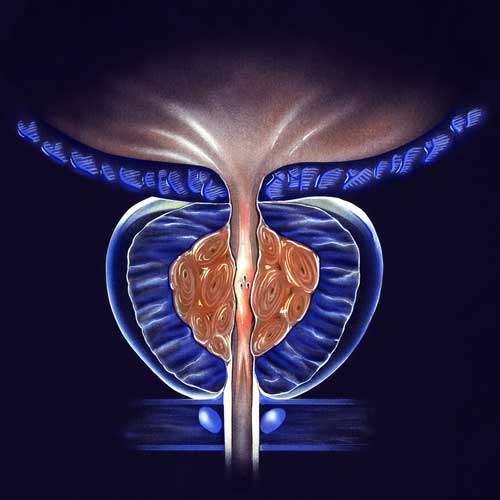
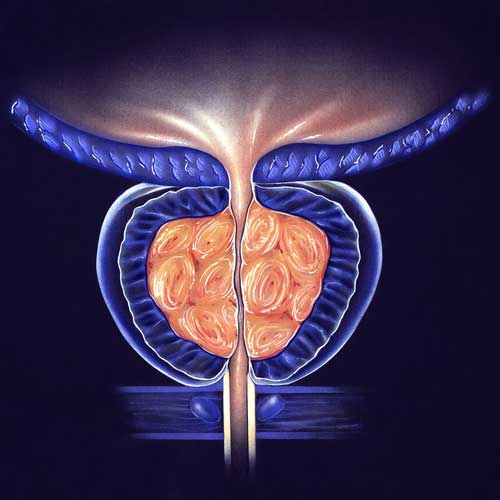
BPH Studies
BPH/ENLARGED PROSTATE CLINICAL TRIALS
A Randomized, International Study to Assess the Safety of iTind Compared to UroLift (MT-08)
With the use of a minimally invasive surgery called UroLift, lower urinary tract symptoms (LUTS) brought on by benign prostatic hyperplasia (BPH) can be relieved. Regarding the improvement of objective and subjective measures, this treatment has demonstrated high efficacy. A temporary device known as the iTind uses minimally invasive surgery to treat LUTS that develops as a result of BPH. Comparing the minimally invasive iTind device to the UroLift device is the goal of this study.
General Requirements:
- Diagnosis of lower urinary tract symptoms presumed to be secondary to benign prostatic enlargement causing bladder outlet obstruction for which treatment is recommended
- Males ≥ 50 years of age or older
- Screening PSA < 4 mg/dl
- Prostate volume up to 75 cc documented by TRUS or MRI.
Customized TULSA-PRO Ablation Registry
Data from patients who have undergone or are currently undergoing the transurethral ultrasound ablation (TULSA) procedure as part of their regular clinical care will be entered into this patient registry. The registry will shed light on actual outcomes of procedure safety and effectiveness and help researchers determine how a patient's quality of life changes over the course of their lifespan and follow-up visits.
General Requirements:
- Male
- >18 years old
- Candidate for TULSA-PRO treatment
- willing and able to sign the Informed Consent form
Contact Us For More Information
Thank you for contacting us.
We will get back to you as soon as possible.
We will get back to you as soon as possible.
Oops, there was an error sending your message.
Please try again later.
Please try again later.
Call 210-617-3670
messages@theUPI.com
9618 Huebner Road, Suite 120
San Antonio, TX 78240
New Patient Referral Fax:
(210) 761-8851
Office Fax: (888) 316-9464
Join Our Newsletter
Join Our N
Thank you for contacting us.
We will get back to you as soon as possible.
We will get back to you as soon as possible.
Oops, there was an error sending your message.
Please try again later.
Please try again later.
Business Hours
- Mon - Fri
- -
- Sat - Sun
- Closed